Market Insights
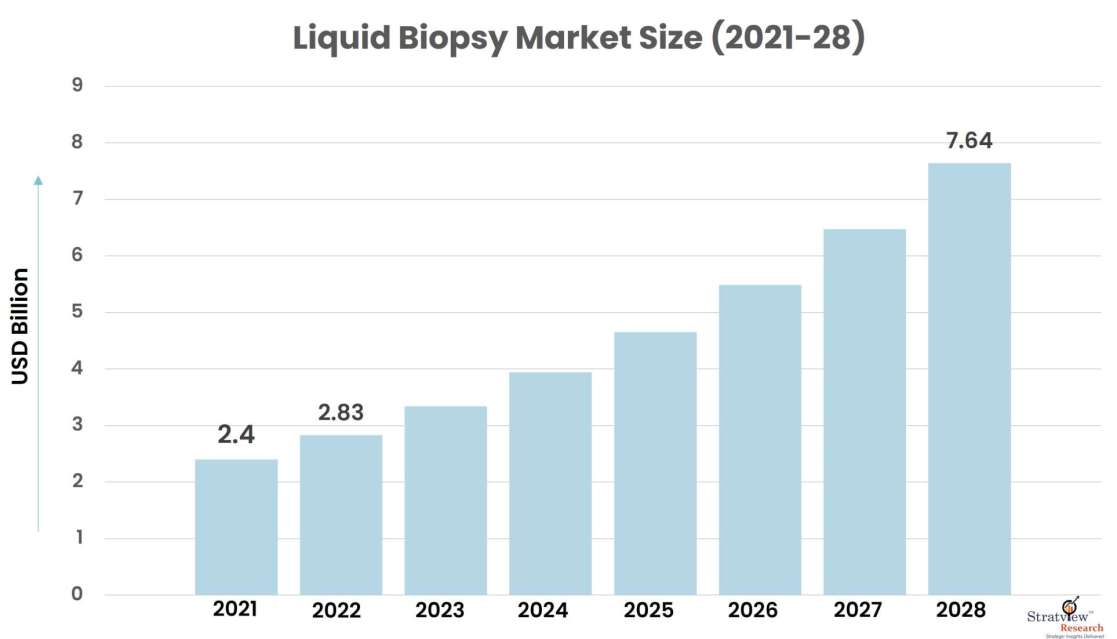
The liquid biopsy market was estimated at USD 2.4 billion in 2021 and is expected to grow at a CAGR of 18% during 2022-2028 to reach USD 7.64 billion in 2028.
Want to know more about the market scope: Register Here
What is a liquid biopsy?
Liquid biopsy, also known as fluid biopsy or fluid phase biopsy, is the sampling and analysis of non-solid biological tissue, mainly blood. This technique is mainly used as a diagnostic and monitoring tool for diseases such as cancer, with the added benefit of being largely non-invasive.
COVID-19 IMPACT
The impact of the global pandemic Covid-19 was felt across all industries in every field. However, the liquid biopsy market managed to escape the pandemic with minimum damage. The liquid biopsy market witnessed a slight decline in the year 2020 during the pandemic and is likely to rebound at a healthy rate in the forecast period.
|
Liquid Biopsy Market Features
|
|
Market Size in 2021
|
USD 2.4 billion
|
|
Market Size in 2028
|
USD 7.64 billion
|
|
Market Growth (2022-2028)
|
18% CAGR
|
|
Base Year of Study
|
2021
|
|
Trend Period
|
2016-2020
|
|
Forecast Period
|
2022-2028
|
|
Segments Covered
|
By Products & Services Type (Assay Kits, Instruments, and Services),
By Application Type (Cancer {Lung Cancer, Breast Cancer, Colorectal Cancer, Prostate Cancer, Melanoma, and Others} and Non-Cancer),
By Clinical Application Type (Therapy Selection, Treatment Monitoring, Recurrence Monitoring, and Early Cancer Screening),
By Circulating Biomarkers Type (Circulating Tumor Cells, Cell-Free DNA, Circulating Tumor DNA, Extracellular Vesicles, and Others),
By Technology Type (Multi-Gene Parallel Analysis Using NGS and Single-Gene Analysis Using PCR Microarrays),
By End-User Type (Reference Laboratories, Hospitals and Physician Laboratories, Academic & Research Centers, and Others),
By Region (North America, Europe, Asia-Pacific, and the Rest of the World).
|
Key Players
Some of the key players in the market are-
- Biocept Inc.
- Bio-Rad Laboratories
- F. Hoffmann-La Roche Ltd.
- Guardant Health Inc.
- Illumina, Inc.
- Johnson & Johnson
- Laboratory Corporation of America Holdings
- MDxHealth SA
- QIAGEN N.V.
- Thermo Fisher Scientific Inc.
Market Drivers
The growth of the liquid biopsy market is primarily driven by-
- Increase in the number of cancer patients,
- Increasing demand for non-invasive treatment procedures,
- Growing awareness about the benefits of liquid biopsy,
- Advantages such as low cost, rapid and accurate results, and early detection of diseases.

Want to have a closer look at this report, Click Here
Segments Analysis
By Application Type
The liquid biopsy market is segmented into cancer and non-cancer applications. Cancer application is further sub-segmented into lung cancer, breast cancer, colorectal cancer, prostate cancer, melanoma, and others. The cancer segment is likely to grow significantly at an exponential CAGR, during the forecast period, largely due to the increasing number of cancer cases and increasing demand for liquid biopsy for diagnosis. In addition, the increasing number of research studies on liquid biopsy for cancer applications is further expected to drive the growth of the market during the forecast period.
By Technology Type
The market is segmented as multi-gene parallel analysis using NGS and single-gene analysis using PCR microarrays. Between these two technology types, the single-gene analysis using the PCR microarrays segment is expected to be dominant in the market over the forecast period. The segment's growth can be attributed to the use of PCR, which is increasing due to its increasing use for various purposes, including detecting fungal infections, bacterial infections, gene mapping, and other genomic applications. PCR tests' quick and real-time diagnostic results are accurate, and they can guide targeted therapies for cancer treatment. PCR is rapidly gaining popularity and is expected to grow at a rapid pace in the coming years.
By End-User Type
Reference laboratories are expected to be the dominant segment of the market during the forecast period.
The market is segmented as reference laboratories, hospitals and physician laboratories, academic & research centers, and others. The outsourcing of liquid biopsy tests to reference laboratories is expected to drive market growth over the forecast period.
Regional Insights
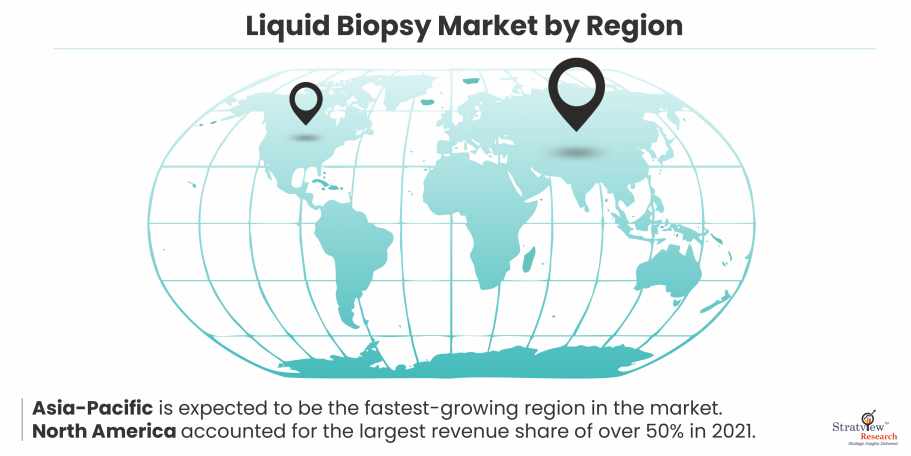
Among these regions, Asia-Pacific is expected to be the fastest-growing region in the market during the forecast period.
In 2021, North America had the highest revenue share of over 50%. Due to higher investments and the existence of numerous biotechnology businesses developing tests in the U.S., this region dominates the most.
The market is segmented into North America, Europe, Asia-Pacific, and the Rest of the world. The region is distinguished by rising disposable income, increasing healthcare expenditure, growing awareness of liquid biopsy techniques, and rising adoption of advanced diagnostic techniques in hospitals, clinics, and diagnostic laboratories. The growing penetration of hospitals and diagnostic centers in the region is likely to bolster the growth of the market over the forecast period. North America and Europe are also expected to offer substantial growth opportunities during the forecast period.
Know the high-growth countries in this report. Register Here
Market Scope & Segmentation
This report studies the market covering a period of 12 years of trend and forecast. The report provides detailed insights into the market dynamics to enable informed business decision making and growth strategy formulation based on the opportunities present in the market.
The market is segmented into the following categories.
By Products & Services Type
- Assay Kits
- Instruments
- Services
By Application Type
By Clinical Application Type
- Therapy Selection
- Treatment Monitoring
- Recurrence Monitoring
- Early Cancer Screening
By Circulating Biomarkers Type
- Circulating Tumor Cells
- Cell-Free DNA
- Circulating Tumor DNA
- Extracellular Vesicles
- Others
By Technology Type
- Multi-Gene Parallel Analysis Using NGS
- Single-Gene Analysis Using PCR Microarrays
By End-User Type
- Reference Laboratories
- Hospitals and Physician Laboratories
- Academic & Research Centers
- Others
By Region
- North America (Country Analysis: the USA, Canada, and Mexico)
- Europe (Country Analysis: Germany, France, the UK, Russia, Spain, and the Rest of Europe)
- Asia-Pacific (Country Analysis: China, Japan, India, South Korea, and the Rest of Asia-Pacific)
- Rest of the World (Sub-Region Analysis: Latin America, the Middle East, and Others)

Click Here, to know the market segmentation details.
Research Methodology
This report offers high-quality insights and is the outcome of a detailed research methodology comprising extensive secondary research, rigorous primary interviews with industry stakeholders and validation and triangulation with Stratview Research’s internal database and statistical tools.
More than 1,000 authenticated secondary sources, such as company annual reports, fact books, press releases, journals, investor presentations, white papers, patents, and articles have been leveraged to gather the data.
We conducted more than 10 detailed primary interviews with the market players across the value chain in all four regions and with industry experts to obtain both qualitative and quantitative insights.
Report Features
This report provides market intelligence in the most comprehensive way. The report structure has been kept such that it offers maximum business value. It provides critical insights into the market dynamics and will enable strategic decision-making for the existing market players as well as those willing to enter the market. The following are the key features of the report:
- Market structure: Overview, industry life cycle analysis, supply chain analysis
- Market environment analysis: Growth drivers and constraints, Porter’s five forces analysis, SWOT analysis
- Market trend and forecast analysis
- Market segment trend and forecast
- Competitive landscape and dynamics: Market share, product portfolio, product launches, etc.
- Attractive market segments and associated growth opportunities
- Emerging trends
- Strategic growth opportunities for the existing and new players
- Key success factors
|
Report Features
|
|
Market Segmentation
|
7
|
|
Regions Covered
|
4
|
|
Countries/Sub-Regions Covered
|
17
|
|
No. of Figures & Tables
|
>150
|
|
Company Profiles
|
10
|
Recent Developments
Jul 2022: Delfi Diagnostics Announces $225 Million Series B Financing to Develop Globally Accessible Portfolio of Liquid Biopsy Tests
Delfi Diagnostics, Inc., a pioneering developer of a new class of high-performance, accessible liquid biopsy tests for early cancer detection and monitoring, today announced a Series B funding of $225 million.
This round of financing was led by DFJ Growth with participation from Eli Lilly and Company, Point72, Brown Advisory, Point Field Partners, Initiate Ventures, Open Field Capital, PTX Capital, and all existing investors including Cowen Healthcare Investments, Foresite Capital, Menlo Ventures, OrbiMed, funds, and accounts advised by T. Rowe Price Associates, Inc., Northpond Ventures, Samsara BioCapital, Rock Springs Capital, AV8 Ventures, Illumina Ventures, Osage University Partners, and Windham Venture Partners.
Jun 2022: Artemis DNA Forays Into Multi-Billion Dollar Cancer Liquid Biopsy Market
The $250 million contract gives Artemis DNA exclusivity to commercialize and distribute Trucheck Pragma and Cancertrack liquid biopsy tests in the USA and Vietnam.
Artemis DNA, Inc. (“Artemis DNA” or the “Company”), a leading USA-based diagnostic laboratory company, announced today that they will be offering revolutionary new cancer liquid biopsy tests through an exclusive partnership with Datar Cancer Genetics (Datar). The agreement, a $250 million contract, grants Artemis DNA the exclusivity to be the only company that will commercialize and distribute the revolutionary cancer liquid biopsy tests, Trucheck Pragma and Cancertrack, in the USA and Vietnam.
Report Customization Options
With this detailed report, Stratview Research offers one of the following free customization options to our respectable clients:
Company Profiling
- Detailed profiling of additional market players (up to 3 players)
- SWOT analysis of key players (up to 3 players)
Market Segmentation
- Current market segmentation by any one of the technology types by application type.
Competitive Benchmarking
- Benchmarking of key players on the following parameters: Product portfolio, geographical reach, regional presence, and strategic alliances
Custom Research: Stratview Research offers custom research services across sectors. In case of any custom research requirement related to market assessment, competitive benchmarking, sourcing and procurement, target screening, and others, please send your inquiry to [email protected].

